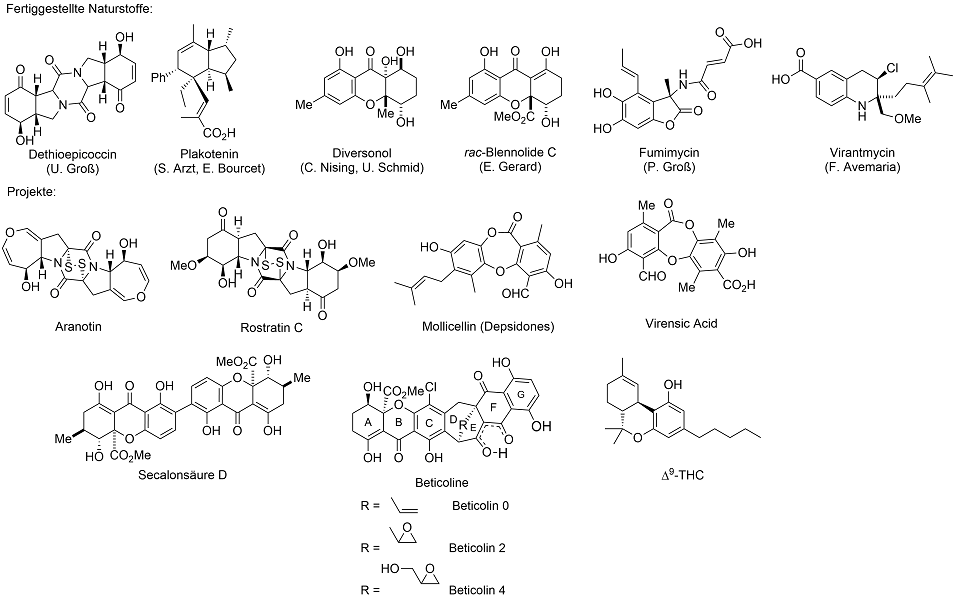
Aranotin
Aranotins belong to a group of mycotoxins with a central epithiodiketopiperazin unit. Aranotin (1a) itself was isolated from the fungus Aspergillus terreus, Acetylaranotin (1b) was additionally isolated from Arachniotus aureus. They show strong antiviral activity in vitro as well as in vivo, especially against polio- (type 1, 2 and 3), coxsackie- (A21), rhino- and parainfluenza viruses (type 1 and 3). The antiviral properties of the metabolites are of special interest for their inhibition of the virus-induced RNA-polymerase without inhibition of the deoxyribonucleic acid-based RNA-polymerase of mammals. Neither Aranotin nor any related dihydrooxepin natural product could be obtained by total synthesis so far.
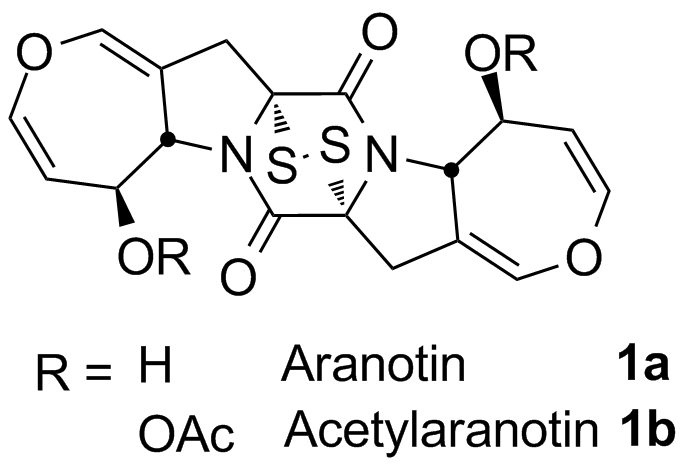
Beticolin 0
Mycotoxins are fungal secondary metabolites exhibiting adverse effects on humans, animals as well as crops, resulting in diseases and economic loss.
Beticolins are mycotoxins produced by the fungus Cercospora beticola which is responsible for the leaf spot disease causing heavy damages to crops worldwide. Furthermore, biological studies revealed a broad cytotoxic profile of beticolins. In order to study their mechanism of action, we are interested in developing synthetic approaches towards the highly complex polycyclic scaffold of these natural products bearing a unique bicyclo[3.2.2]nonane ring system.
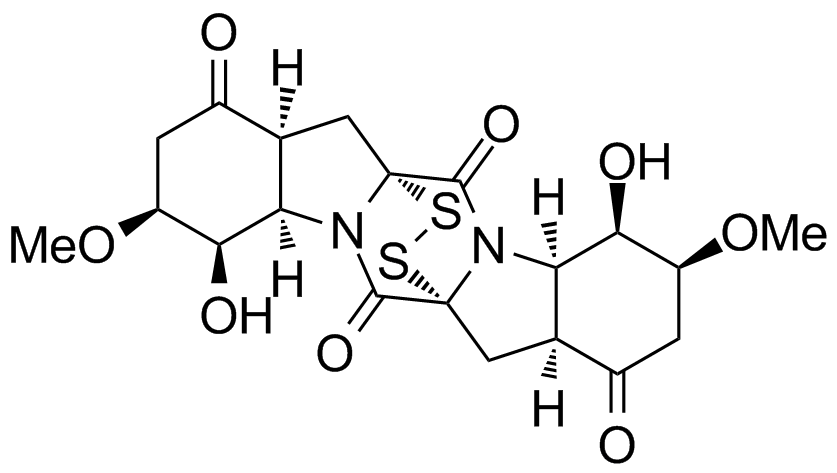
Rostratin C
The rostratines A–D belong to the class of mycotoxines. The marine natural products with the epithiodiketopiperazin unit were isolated from the fungus Exerohilum rostratum close to Hawaii in 1997. In 2004 the structures of all four secondary metabolites could be elucidated. Their in vitro cytotoxicity against the human colorectal cancer of the cell line HCT-116 make them interesting as new lead structures for the natural product synthesis.
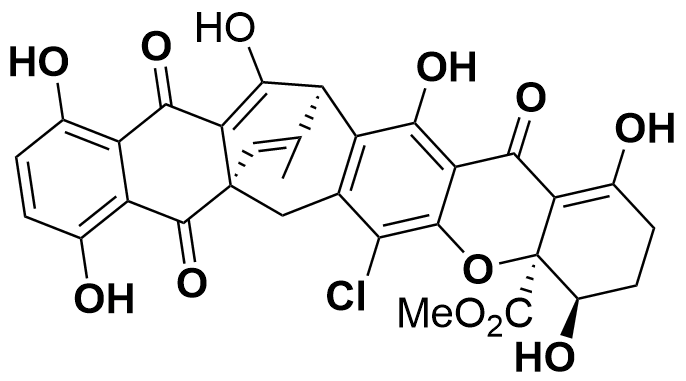
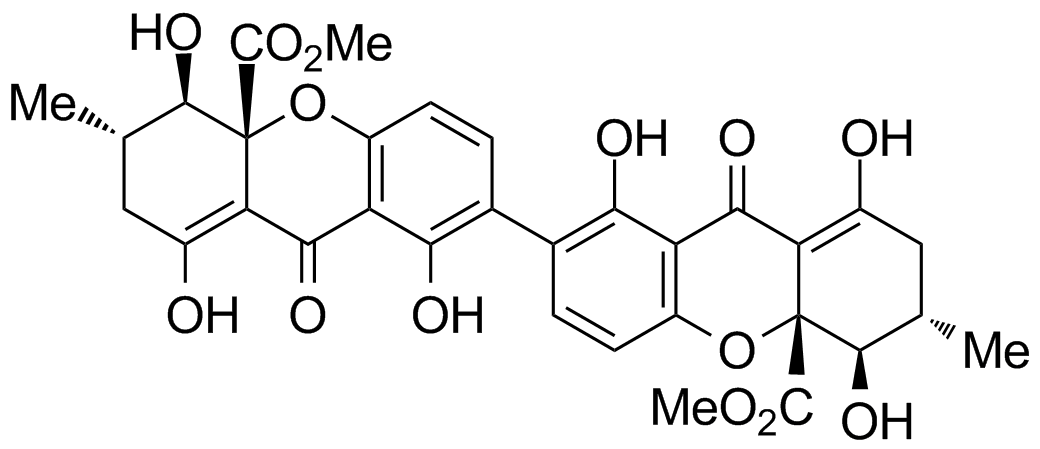
Secalonsäure D
The secalonic acids A-G were isolated for the first time from the ascomycotete Claviceps purpurea (“ergot”) and show versatile biological activity, e.g. cytotoxicity and antibacterial properties. They were discovered more than 100 years ago, still no total synthesis of this compound class has been achieved until today.
Spiculoinsäure A
In 2004 the previously unknown polyketidic natural product spiculoic acid A was isolated from the Caribbean sponge Plakortis angulispiculatus (Carter) by Andersen et al. The structure and relative stereochemistry of the highly substituted hydrindane was elucidated by a combination of mass spectrometry and NMR studies. Further on the substance shows cytotoxic activity against the human breast cancer cell line MCF-7 (IC50 8 µg/ml), which makes it an interesting candidate for drug design.
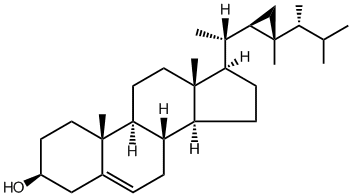
Gorgosterol
Gorgosterol and a wide variety of other sterols are produced by dinoflagellates, including those in the genus Symbiodinium, the obligate intracellular symbionts of reef-building corals. To date the biological functions of gorgosterol and other sterols in this symbiosis are unknown; we hypothesize that they are transferred from the symbiont to the coral host where they may play an important role in lipid metabolism or steroid signaling. We aim to synthesize gorgosterol, potentially also with modifications, to assess its biological functions in symbiosis using the marine sea anemone Aiptasia, an emerging model system for corals.
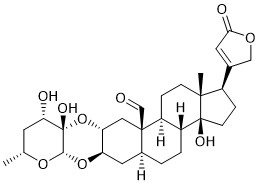
Calotropin
The naturally occurring steroid Calotropin belongs to the class of cardiac glycosides, which have been used as therapeutics for the treatment of congestive heart failure for over 200 years. In recent years, their emerging role as potential anticancer agents was revealed. Calotropin in particular has been identified as inhibitor of the Wnt signaling pathway. This pathway regulates a number of important cellular processes, including embryonic development, cell differentiation and proliferation and can also be associated with various human diseases. For instance, 80% of colon cancer cases can be attributed to an aberrant active Wnt signaling pathway. Inhibitors of this pathway, such as Calotropin, could therefore serve as lead structures for new therapeutics in Wnt signaling-dependent cancer. Not only the interesting biological properties, but also the synthetically challenging structure of Calotropin have motivated us to develop a strategy for its semisynthesis starting from readily available starting materials like methyl α-D-glucopyranoside and epi‑androsterone. For the synthesis of the ulosyl bromide a literature known strategy is followed, while the synthesis of Calotropagenin starting from epi-androsterone comprises the following aspects:
- Oxidation of C-2 and C-19 to enable introduction of the trans 2α,3b-diol structural motif and aldehyde functionality
- Diastereoselective introduction of the b-hydroxyl group under inversion of the present stereogenic center
- Introduction of the γ-butenolide ring at C-17