Copper-Based TADF Metal Complexes
Luminescent copper(I) complexes present a promising class of emitting materials for organic light-emitting diodes (OLEDs) and have attracted enormous interest due to their high emission efficiencies, color tunability by ligand variation, and their abundancy compared to iridium(III) or platinum(II) compounds. Especially the assessment of thermally activated delayed fluorescence (TADF) to copper(I) compounds has accelerated the development and investigation of several complex classes, and ligand (and halide) variation helped to understand the fundamental photophysical properties of the different coordination motifs.[1]
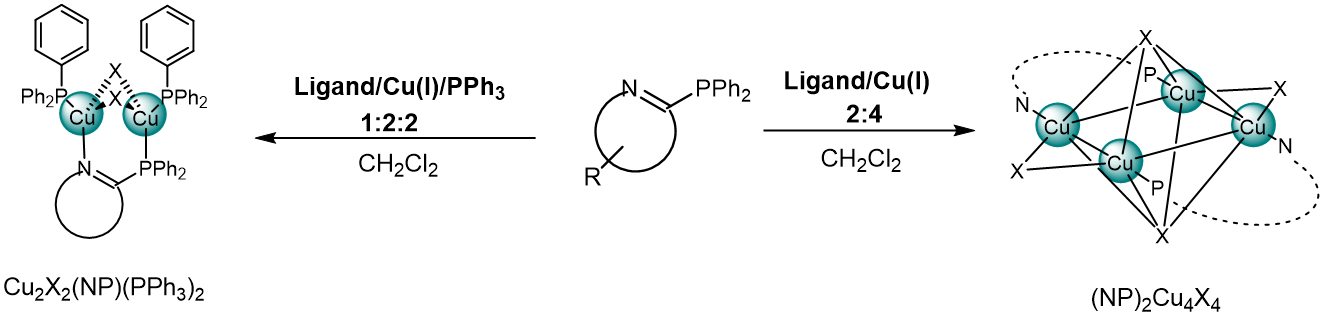
Figure 1. Synthesis of dinuclear and tetranuclear Cu(I)-NHetPHOS complexes.
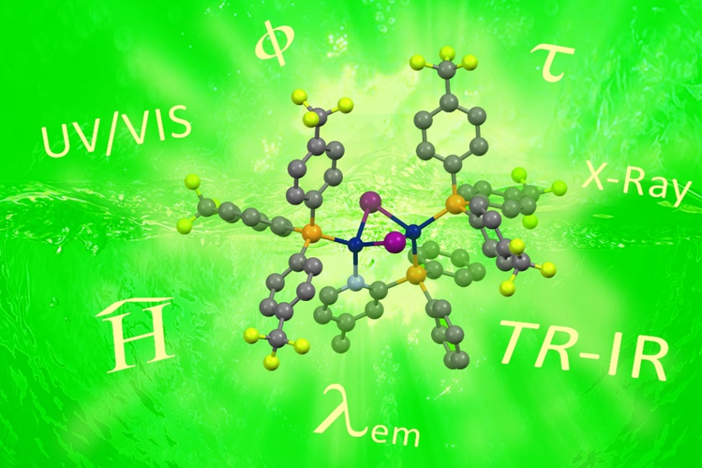
Figure 2. The Cu(I)-NHetPHOS complexes are investigated by a combination of the photophysical studies, the time-resolved step-scan FTIR technique and theoretical calculations.
NHetPHOS (N-heterocyclic phosphine)-type copper complexes with the butterfly-shaped Cu2X2 core lead to a rigid structure, which generally lead to improved photophysical properties.[2] The color of the emitters can be tuned by introducing electronic groups on the bridging ligand.[3] The solubility can be modified by introducing other appropriate substituents.
Compared with metal mononuclear complexes, multinuclear complexes with richness of the geometry motifs allow combining with significant metal-to-ligand charge transfer (MLCT) contribution and appreciable spin-orbit coupling (SOC), which is crucial for a highly efficient TADF emission.[4]
In general, the synthesis of Cu(I) complexes is carried out at room temperature by mixing solutions of a metal precursor and one or more ligands. In many cases, the reaction turnover is nearly quantitative and high yields may be achieved. Photoluminescence spectroscopy, TCSPC (time-correlated single photon counting), cyclic voltammetry, X-ray diffraction, and DFT calculations were performed to gain a broad understanding of the emissive cooper complexes.[5]
Figure 3. (a) Scheme of highly soluble Cu(I)-NHetPHOS complex inkjet processing (b) Yellow-green emission from an OLED prototype with dinuclear Cu(I)-NHetPHOS complex as a luminophore.
Inkjet printing is a versatile digital printing technique and often used for fabricating OLEDs with low environmental impact. Almost all printing techniques offer roll-to-roll fabrication thus high volume and high-speed production. Furthermore, a broad variety of materials can be used as substrates: for example, paper can serve as a carrier for low scale printed electronic devices. By using a well-soluble Cu(I)-NHetPHOS emitter, our group were able to realize an inkjet-printed OLED, where the emitting layer was printed under ambient conditions. In the refined device, we yielded efficiency values of more than 11% EQE and ultrahigh brightness values of 34 000 cd m-2. [6]
[1] D. Volz, D. M. Zink, T. Bocksrocker, J. Friedrichs, M. Nieger, T. Baumann, U. Lemmer, S. Brase, Chem. Mater. 2013, 25, 3414–3426.
[2] D. Volz, Y. Chen, M. Wallesch, R. Liu, C. Fléchon, D. Zink, J. Friedrichs, H. Flügge, R. Steininger, J. Göttlicher, C. Heske, L. Weinhardt, S. Bräse, F. So, T. Bauman, Adv. Mater. 2015, 27, 2538–2543.
[3] Daniel M. Zink, T. Baumann, J. Friedrichs, M. Nieger, S. Bräse, Inorg. Chem. 2013, 52, 13509–13520.
[4] C. Bizzarri, E. Spuling, D.M. Knoll, D. Volz, S. Bräse, Coord. Chem. Rev. 2018, 373, 49–82.
[5] ] J. M. Busch, D. M. Zink, P. Di Martino-Fumo, F. R. Rehak, P. Boden, S. Steiger, O. Fuhr, M. Nieger, W. Klopper, M. Gerhards, S. Bräse, Dalton Trans. 2019, 48, 15687–15698.
[6] M. Wallesch, A. Verma, C. Flechon, H. Flegge, D. Zink, S. Seifermann, J. Navarro, T. Vitova, J. Göttlicher, R. Steininger, L. Weinhardt, M. Zimmer, M. Gerhards, C. Heske, S. Bräse, T. Baumann, D. Volz, Chem. Eur. J. 2016, 22, 16400–16405.