Synthesis of new fluorophores for biological imaging
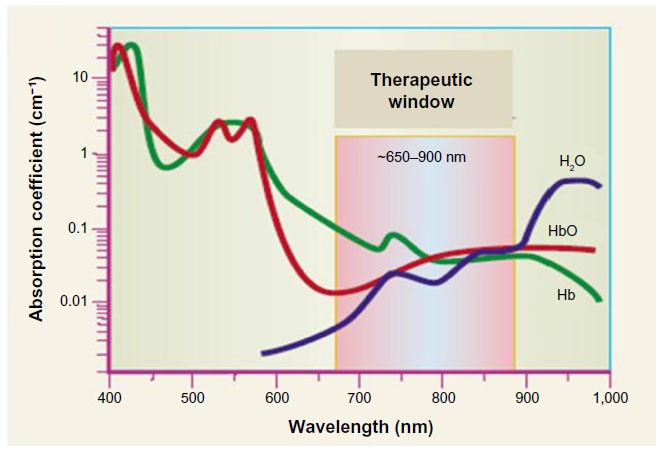
Optical imaging has a potential to timely diagnose various diseases, such as, for instance, different types of cancer, as well as to create personalized "road maps" for their treatment. The search for the new luminescent materials which can be implemented as efficient biomarker is a crucial task nowadays. Among all the fluorophores, the most promising candidates are those which have an emission in red of infrared region, because the living tissue transmission is sufficiently large and the scattering is low in red-infrared region. As for red-emitters, europium-based coordination compounds with their metal-centered red emission (612-615 nm) is one of the promising classes of such materials due to the long lifetimes of the excited states and large Stokes shift, quantum yields up to 80% which could be also used for time-resolved microscopy. Among NIR- and IR emitters, pyrrolopyrrolcyanine dyes have unique photo physical properties. On that account they are of great interest for biological imaging experiments.
Lanthanide-based compounds and their conjugates with molecular transporters
Lanthanides, namely the elements from Ce to Lu are unique among other elements. The shielding of the 4f orbitals result in special spectroscopic properties with parity-forbidden f–f absorptions having very low molar extinction coefficient and characteristic narrow-line emission. The problem of poor f-f absorption is resolved by chromophore ligand, complexing the metal, which is called antenna effect.
The current research is devoted to the design of new Ln-based luminescent probes with improved photophysical propties to obtain fluorophores for advanced biological applications (such as time-resolved microscopy or FRET-bioassay). The research project consists of 3 important milestones, namely (i) the optimization of the structure of Ln—based compound in terms of stability, solubility and luminescence; (ii) modification of the compound with linkers, which is in fact the preparation of the probe for further comjugation and (iii) the conjugation of the ln-based probe with molecular transporters.
Different classes of Ln-based compounds can be investigated. For instance, we have shown, that Eu fluorobenzoates are suitable for cellular imaging and ink-jet printing [1,2].
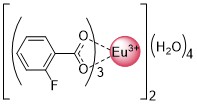
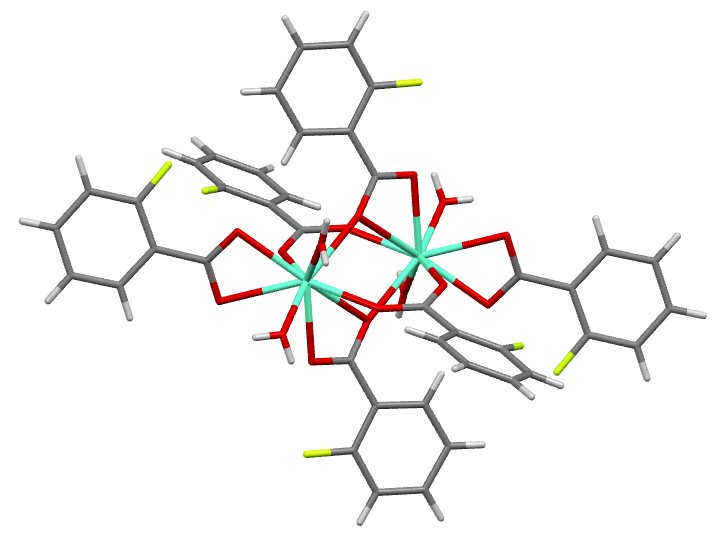
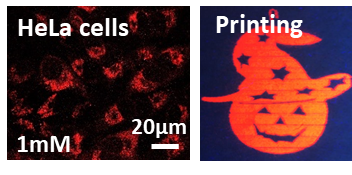
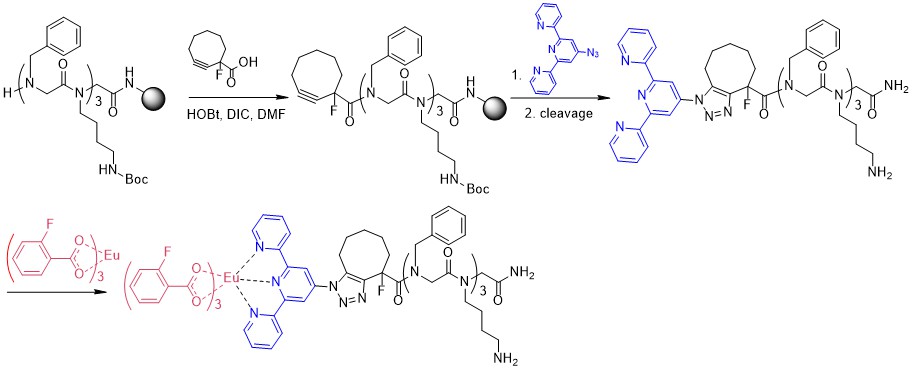
We also developed several approaches to increase the efficiency of lanthanide-based probes.
- The introduction of additional metal as an additional sensitization pathway [1,2]
- The introduction of additional ligand as an additional absorbing and sensitizing part [3]
Lanthanide complexes may be linked with peptoids using coordination bonds due to the strong coordination of lanthanides to 1,10-Phenanthroline moieties and its derivatives
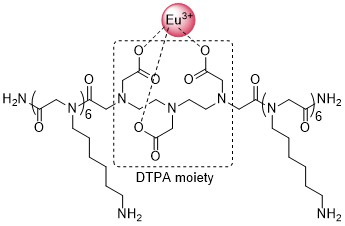
 Alternatively, chelating moiety (e.g. DTPA) might be incorporated into the peptoid structure [4]
Alternatively, chelating moiety (e.g. DTPA) might be incorporated into the peptoid structure [4]
List of publications:
[1] Kalyakina A.S., Bräse S. et al. Eur. J. Chem. 2015, 21, 17921-32
[2] Kalyakina A.S., Bräse S. et al. Eur. J. Chem. 2017, 23, 14944-53.
[3] Kalyakina A.S., Bräse S. et al. Chem. Commun. 2018, 54, 5221-4.
[4] Kölmel D. K., Bräse S. et al. Eur. J. Org. Chem. 2013, 2761–2765.
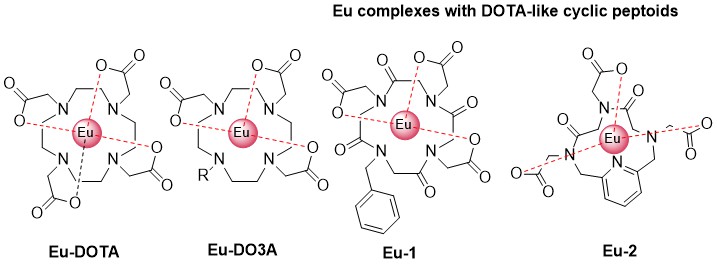
DOTA-like cyclic peptoids and Ln complexes with them
Among the diverse properties of cyclic peptoids used as peptidomimetics, their ability to coordinate different metal ions has recently been reported. The presence of multiple donor atoms makes it easy to coordinate the lanthanide (III) ion within the cyclic peptoid. Additional solubility in water, which is difficult to achieve for many lanthanide complexes, gives such a compound great potential for many biological applications. The chelating complexing agent DOTA, which has a high affinity to trivalent lanthanide ion, has become widely used as a radiopharmaceutical in therapy and diagnostics, e.g. as MRI contrast agent. In the present work the library of DOTA-like peptoids with carboxylic functionality was synthesized. To ensure the antenna effect, the sensitizing part as π-conjugated system such as a benzene ring was introduced as a peptoid side chain. Another way to introduce sensitizer is the use of heterocyclic moieties during the peptoid synthesis, which gives and additional coordinating sites inside the cycle, promoting the coordination of the lanthanide ion inside the peptoid cavity. Thus, two types of DOTA-like cyclic peptoids were investigated as well as Eu(III) complexes with them.