Molecular photoswitches
Molecular photoswitches are "adapters" that reversibly convert light into changes of molecular parameters (geometry, polarity, stiffness). These changes, in turn, may induce modulation of bioactivity, catalytic efficiency, or macroscopic properties of materials.
Established molecular switches (azobenzenes, diarylethenes, spiropyrans) have been recently complemented with new scaffolds (indigoids, arylhydrazones, DASA). Our laboratory is active in the area of new biocompatible azobenzene analogues. We also discovered and explore novel peptide-derived photoswitches - hemipiperazines.
[1] review - Pianowski Z. Recent implementations of molecular photoswitches into smart materials and biological systems. Chem. Eur. J., 2019, 25, 5128-5144
https://onlinelibrary.wiley.com/doi/10.1002/chem.201805814
[2] book - "Molecular photoswitches. Chemistry, properties and applications" (Wiley-VCH, Weinheim 2022, Ed. Z. Pianowski)
https://onlinelibrary.wiley.com/doi/book/10.1002/9783527827626
[3] review - Leistner A.-L., Pianowski Z. Smart photochromic materials triggered with visible light Eur. J. Org. Chem., 2022, 19, e202101271
https://doi.org/10.1002/ejoc.202101271
[4] bookchapter - Pianowski Z. Non-established photoswitchable organic systems RSC Specialist Periodical Reports: Photochemistry, 2023, 51, 434–452
https://doi.org/10.1039/BK9781837672301-00436
Supramolecular photochromic hydrogels
Numerous amphiphilic small molecules form hydrogels in contact with water. Amphiphilic cyclic dipeptides are known as good supramolecular hydrogelators. Responsive hydrogels are important for applications like controlled release of drugs, 3D cell cultures, or construction of adaptive materials. Based on the fact that polarity of azobenzenes can be reversibly changed with light, we designed several photochromic supramolecular hydrogelators based on the cyclic dipeptide scaffold. They form hydrogels under aqueous physiological conditions, and these materials can be reversibly photoliquefied to non-viscous fluids,[5-7] or contracted upon irradiation with concomitant ejection of encapsulated cargo.[8]
The hydrogelators contain unnatural aminoacids bearing an azobenzene photoswitch as the light-sensitive motif. They form self-healing supramolecular hydrogels in aqueous solutions under physiological conditions, which properties can be modified by changing the ionic strength or acidity.
The hydrogels could release previously encapsulated cargo (like long dsDNA, or anticancer drug doxorubicin) in the light-controlled maner, upon exposure to UV,[5] green,[6-7] or red light.[8]

Supramolecular hydrogel formed from PAP-DKP-Lys in water (left) becomes liquid upon UV light irradiation (right). The gel is reconstituted after irradiation with blue light. [5]
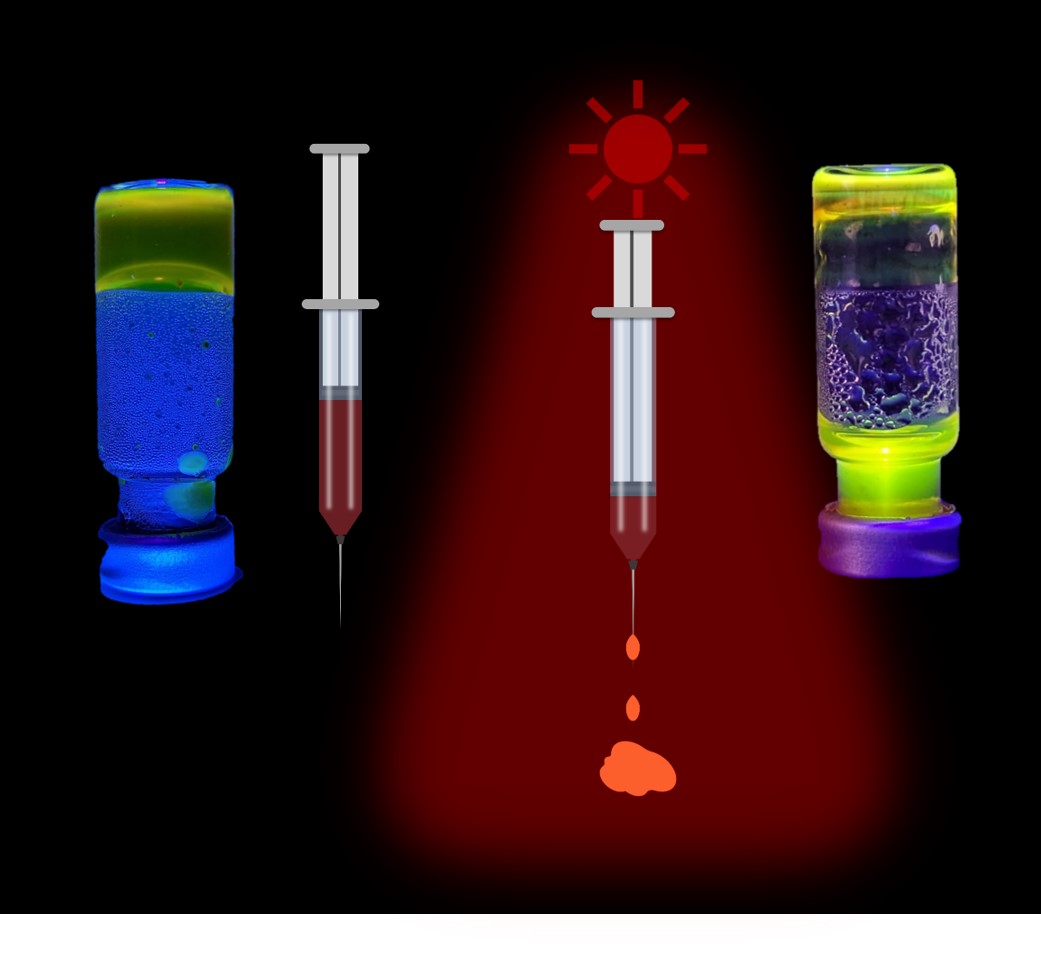
"Molecular syringe" - the hydrogel under physiological conditions shrinks upon exposure to red light (660nm). The cargo (here fluorescein) is concomitantly ejected as non-viscous solution. The material can be applied for photocontrolled drug release inside soft human tissues, which can be well penetrated with red light.[8]
Later on, we discovered that our basic, positively charged hydrogelators can form composite hydrogels with oppositely charged alginate polymers (polyacids). The resulting materials also reversibly liquefy upon irradiation, but require much lower input of the photochromic component to achieve comparable parameters. They can also be prepared upon mixing of the components (as stock solutions) followed by light-driven gelation at room temperature. And thus enable encapsulation of heat-sensitive cargo (proteins, live cells) without temperature-driven degradation or denaturation.[9-10]
[5] Z. Pianowski, J. Karcher, K. Schneider Photoresponsive self-healing supramolecular hydrogels for light-induced release of DNA and doxorubicin Chem. Commun., 2016, 52, 3143-3146.
https://doi.org/10.1039/C5CC09633B
[6] Karcher J., Pianowski Z. Photocontrol of Drug Release from Supramolecular Hydrogels with Green Light Chem. Eur. J., 2018, 24, pp. 11605-11610
https://doi.org/10.1002/chem.201802205
[7] Karcher J., Kirchner S., Leistner A.-L., Hald C., Geng P., Bantle T., Gödtel P., Pfeifer J., Pianowski Z. Selective release of a potent anticancer agent from a supramolecular hydrogel using green light RSC Adv., 2021, 11, 8546-8551
https://doi.org/10.1039/D0RA08893E
[8] Leistner A.-L., Most M. M., and Pianowski Z. Molecular syringe for cargo photorelease - red-light-triggered supramolecular hydrogel Chem. Eur. J. 2023, e202302295
https://doi.org/10.1002/chem.202302295
[9] Leistner A.-L., Kistner D. G., Fengler C., Pianowski Z. Reversible photodissipation of composite photochromic azobenzene-alginate supramolecular hydrogels RSC Adv., 2022,12, 4771-4776
https://doi.org/10.1039/D1RA09218A
[10] Seliwjorstow A., Bach M., Kirchner S., Palloks S., Pianowski Z.L. Visible Light-Triggered Supramolecular Hydrogel Based on Cyclic Dipeptides Stabilized with Coulomb Interactions Macromol. Mater. Eng. 2024, 2400007
https://doi.org/10.1002/mame.202400007
Hemipiperazines (HPI)
In 2022, we were first to report [11] that arylidene substituents on 2,5-diketopiperazine (DKP, "glycine anhydride", cyclic dipeptide motif) can be photoisomerized between the thermally stable Z-isomer and the E-isomer (which in most cases shows large thermal stability as well, and thus can be considered metastable at ambient temperatures). The novel photoswitch was named "hemipiperazine" or "HPI" - an abbreviation from "hemistilbene-diketopiperazine".
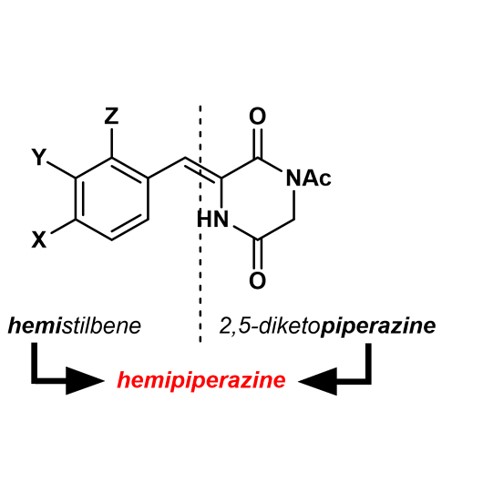
HPI was initially identified [11] in the structure of plinabulin (Z-1) - a low-nM inhibitor of microtubule dynamics and an anticancer drug candidate in 3rd phase of FDA clinical trials against NSCLC and other tumors.
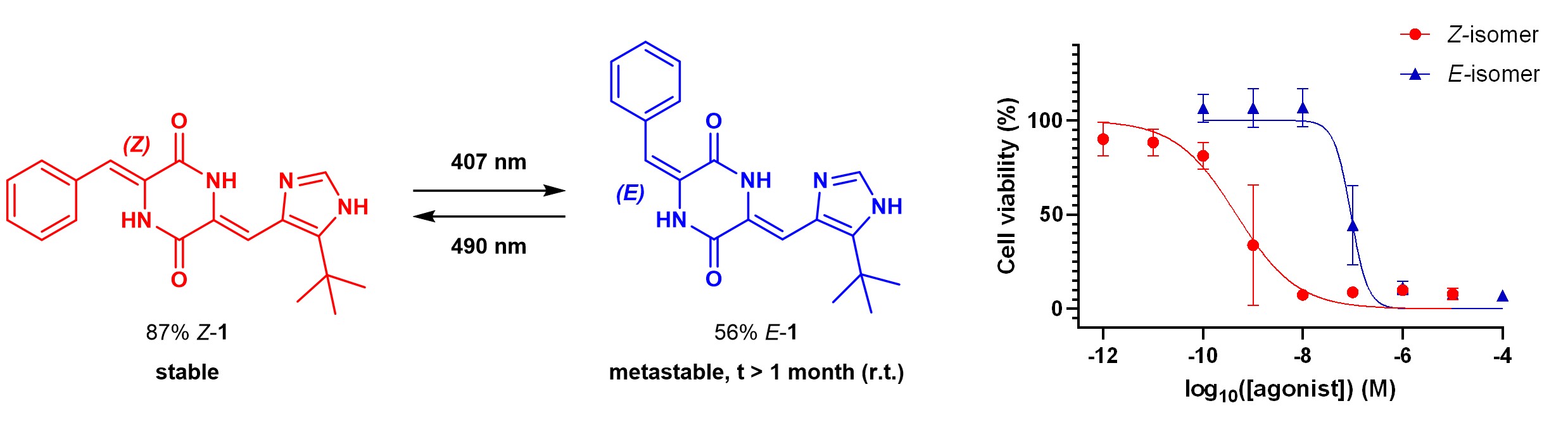
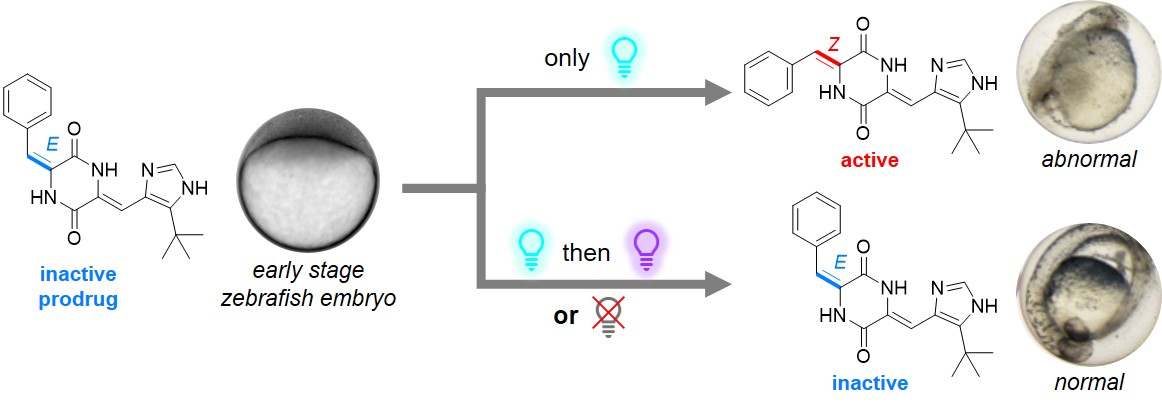
Plinabulin can be irradiated and photoisomerizes to E-1, which is thermally metastable, can be isolated, and its cytotoxicity is lower by two orders of magnitude. The low-nM activity can be restored upon irradiation with cyan or blue light (450-490 nm). We have successfully used this effect to control microtubule-driven early development stage (epiboly) in zebrafish embryos.[14]
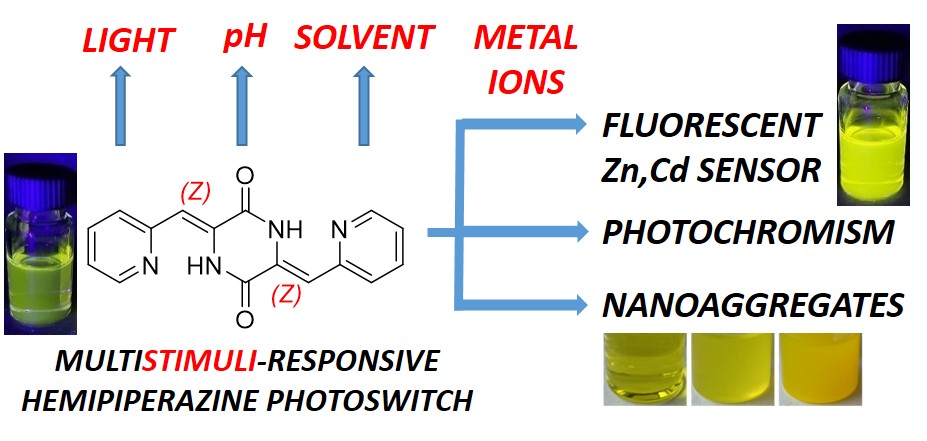
Later, we determined the scope of photochromism for a representative set of carbocyclic HPIs [11] and heterocyclic HPIs.[12] Among the later ones, bis-pyridine-substituted DKP became a photoswitchable fluorescent sensor for Cd and Zn ions.[13]
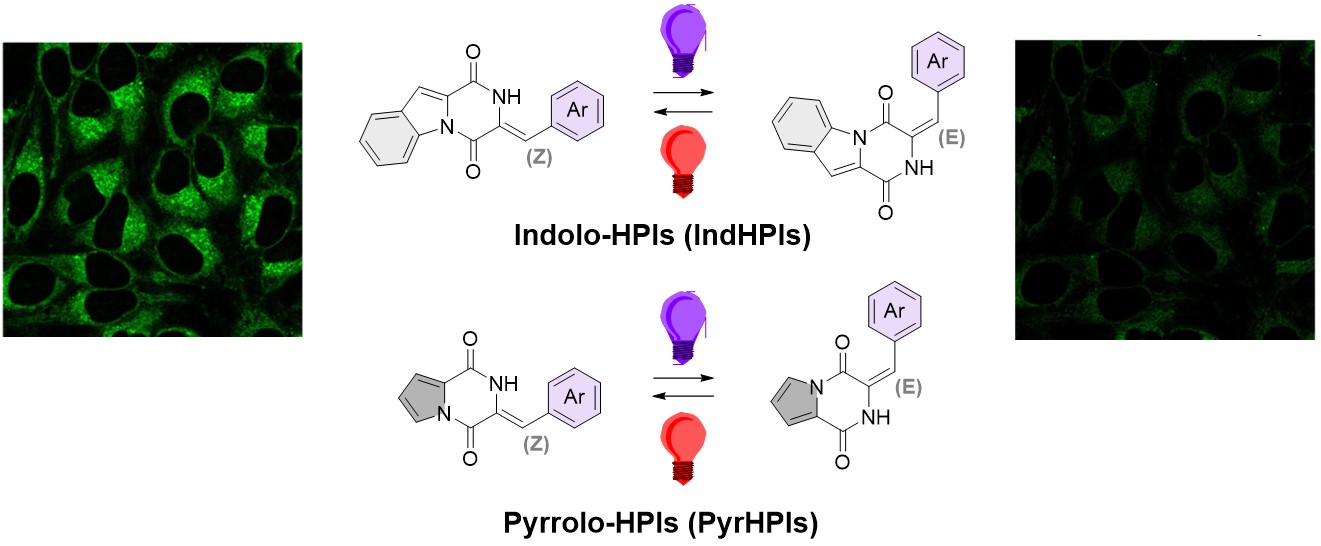
Finally, indole-derived HPIs ("locked plinabulin" system) were identified as photoswitchable fluorophores.[11] HPI as peptide-derived switches with high biocompatibility are intensively explored in our group for preparation of novel biocompatible responsive materials, and in photopharmacology.
We have also explored indolo-HPIs and pyrrolo-HPIs (derivatives of the "locked" plinabulin) as photoswitchable fluorophores reversibly triggered with visible light frequencies, also inside living mammalian cells.
[11] Kirchner S., Leistner A.-L., Gödtel P., Seliwjorstow A., Weber S., Karcher J., Nieger M., Pianowski Z. Hemipiperazines as peptide-derived molecular photoswitches with low-nanomolar cytotoxicity. Nat. Commun. 2022, 13, 6066
https://doi.org/10.1038/s41467-022-33750-7
[12] Gödtel P., Starrett J., Pianowski Z. Heterocyclic Hemipiperazines: Water-Compatible Peptide-Derived Photoswitches Chem. Eur. J. 2023, chem.202204009
https://doi.org/10.1002/chem.202204009
[13] Schäfer V., Pianowski Z. Heterocyclic Hemipiperazines: Multistimuli-responsive Switches and Sensors for Zinc or Cadmium Ions Chem. Eur. J. 2024, e202402005
https://doi.org/10.1002/chem.202402005
[14] Seliwjorstow A., Takamiya M., Rastegar S.*, Pianowski Z.* Reversible Influence of Hemipiperazine Photochromism on the Early Development of Zebrafish Embryo ChemBioChem 2024, e202400143.
https://doi.org/10.1002/cbic.202400143
[15] Gödtel P., Rösch A., Kirchner S., Elbuga-Ilica R., Seliwjorstow A., Fuhr O., Schepers U.*, and Pianowski Z.*
Photoswitchable Fluorescence of Peptide-Based Hemipiperazines Inside of Living Cells
J. Am. Chem. Soc. 2025 147(30), 26652-26662
https://doi.org/10.1021/jacs.5c07013
Novel biocompatible photoswitches
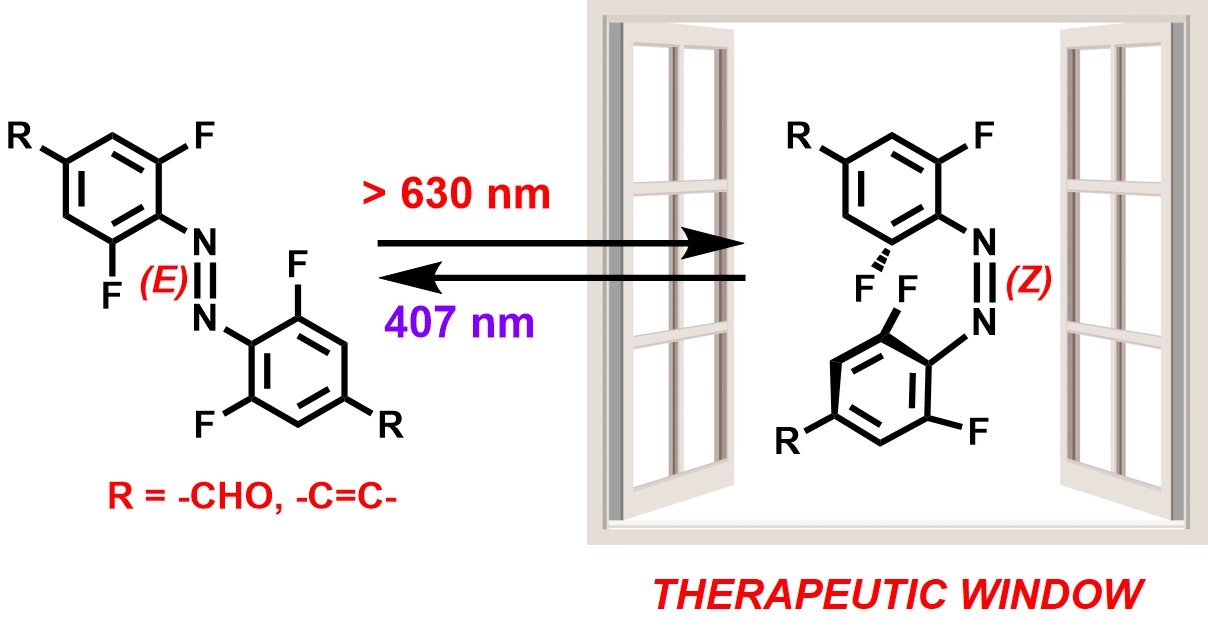
Therapeutic window is the range of light frequencies (c.a. 630-900 nm, red and near-IR light) which can efficiently penetrate soft human tissues and may be used for general human phototherapies (e.g. against solid tumors). Therefore, for broad therapeutic applications of photoswitchable antibiotics or anticancer agents it is important to search for molecular photoswitches that can be activated within that range of light.
We reported that fluorinated azobenzenes (usually isomerized with green light, 500-530 nm) can be triggered with red light (>660 nm), once their p-electron system is sufficiently extended (by adding aldehyde groups or C=C bonds in conjugated positions).[16] These switches may be then integrated with pharmacophores for photopharmacology in vivo.
In collaboration with Dr. Bartosz Lewandowski (ETH Zürich), we developed photoswitchable molecular tweezers triggered with red and violet light, as receptors for basic aminoacid derivatives and biogenic amines, operational in water and physiological buffers (PBS).[17]
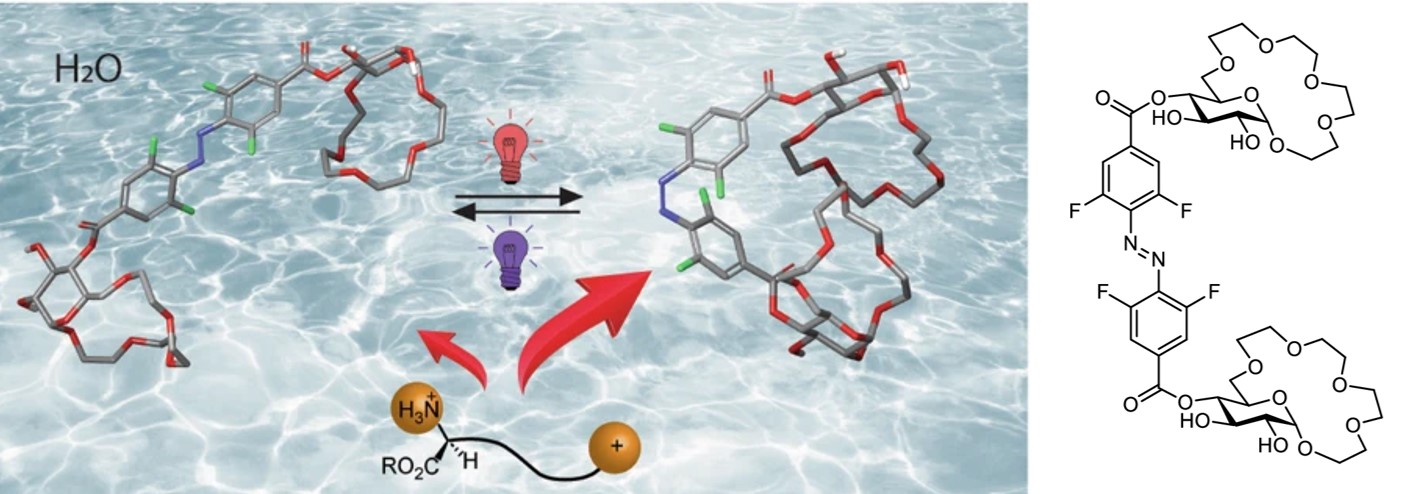
Our group also developed photoswitchable molecular cages, synthesized via dynamic covalent chemistry. The cages can reversibly photoisomerize in water, triggered with green or red light.[18]
[16] Leistner A.-L., Kirchner S., Karcher J., Bantle T., Schulte M. R., Gödtel P., Fengler C., Pianowski Z. Fluorinated Azobenzenes Switchable with Red Light
Chem. Eur. J., 2021, 27(31), 8094-8099.
https://doi.org/10.1002/chem.202005486
[17] Most M.M., Boll L.B., Gödtel P., Pianowski Z.L.*, Lewandowski B.*
Glucose-derived receptors for photocontrolled binding of amino acid esters in water
Commun. Chem. 2025, 8, 50
https://www.nature.com/articles/s42004-025-01445-x https://rdcu.be/eaDl9
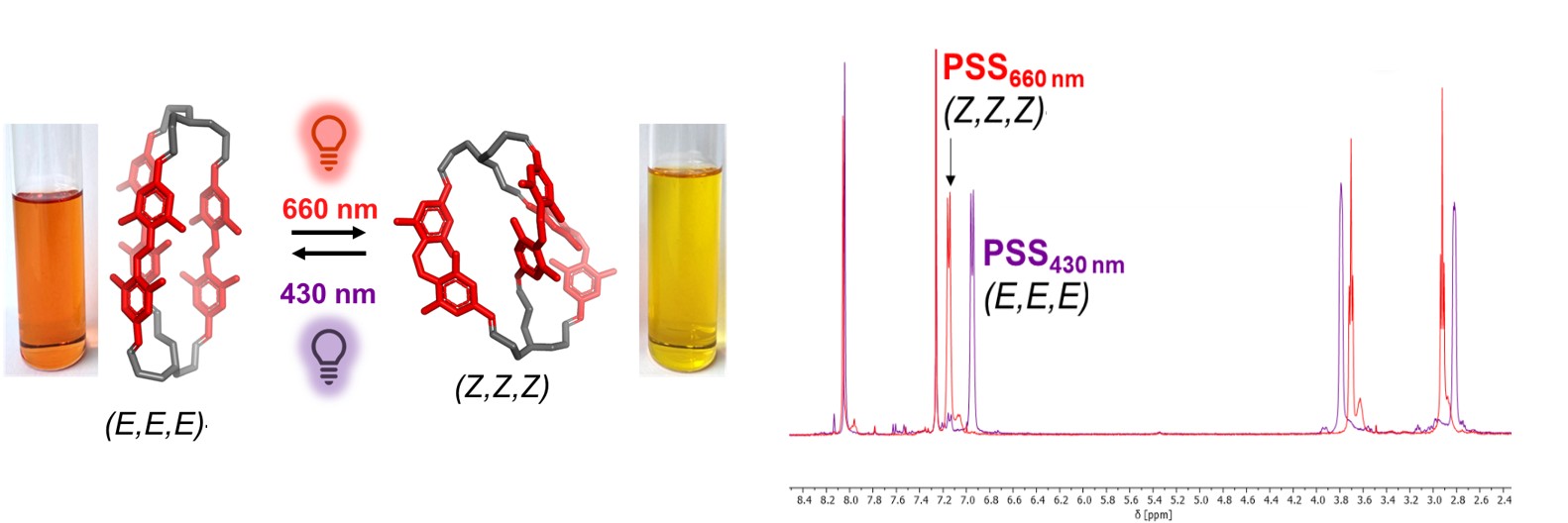
[18] Schäfer V., Seliwjorstow A., Fuhr O., Pianowski Z.L.*
Dynamically assembled photochromic cages operational in water with visible light.
Nat. Commun. 2026, 17, 2488
https://doi.org/10.1038/s41467-026-70406-2 https://rdcu.be/e8LTy

