Organo-Catalysis
Asymmetric Strecker-Reaction with Quinine
An efficient, organocatalytic enantioselective synthesis of N-arylsulfonyl α-amino nitriles from the corresponding α-amido sulfones has been developed. This quinine-catalyzed Strecker reaction provides the corresponding cyanated products in good yields and enantioselectivities (Scheme 1).
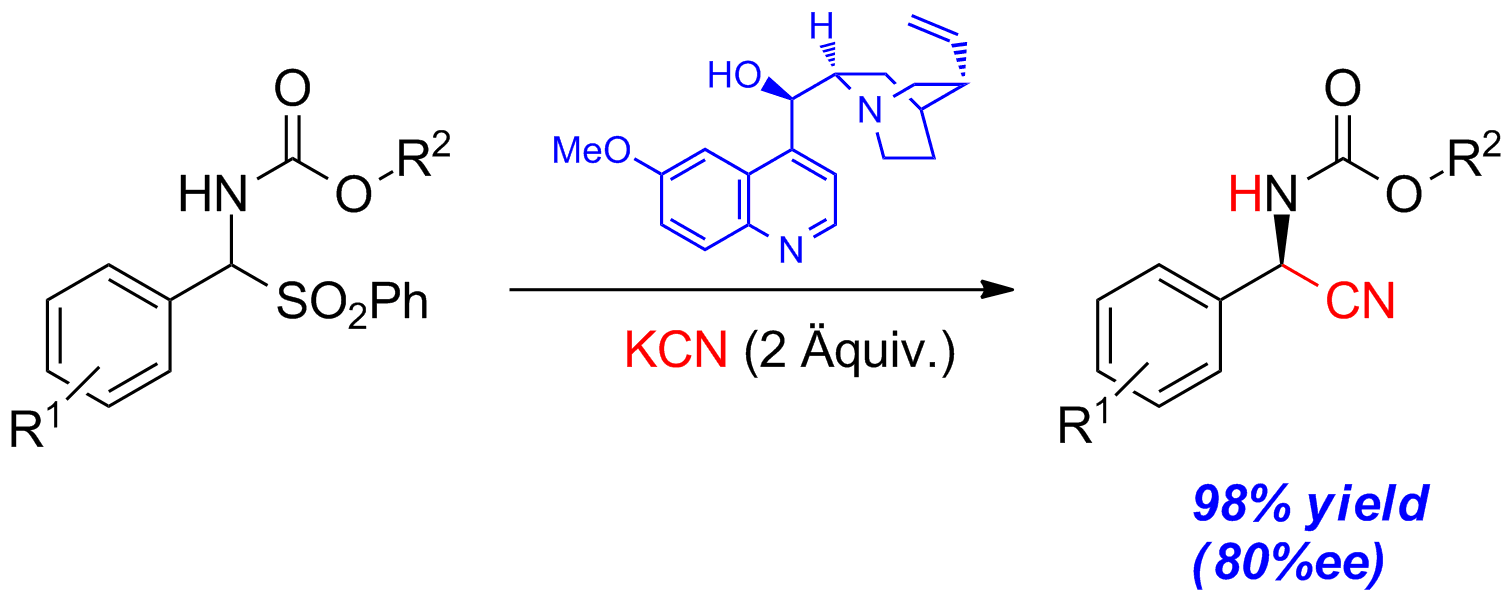
Scheme 1: Organocatalytic Strecker reaction with quinine.
To avoid the highly toxic and volatile HCN, we used potassium cyanide (KCN), a more convenient cyanation reagent, which is easier to handle. In this process, KCN plays two roles. It first acts as a base, liberating the free N-carbamoyl imine by deprotonation of precursor 1 and subsequent elimination of the sulfinate. The intermediately formed HCN then acts as the cyanating agent giving enantioenriched product 3 in the presence of suitable chiral catalysts.
Asymmetric Amination of Aldehydes
An efficient access to configurationally stable α,α-disubstsituted α-amino acids is highly appealing. Starting from simple and easily available racemic aldehydes, the α-sulfamidated could be obtained in good yields and enantioselectivities.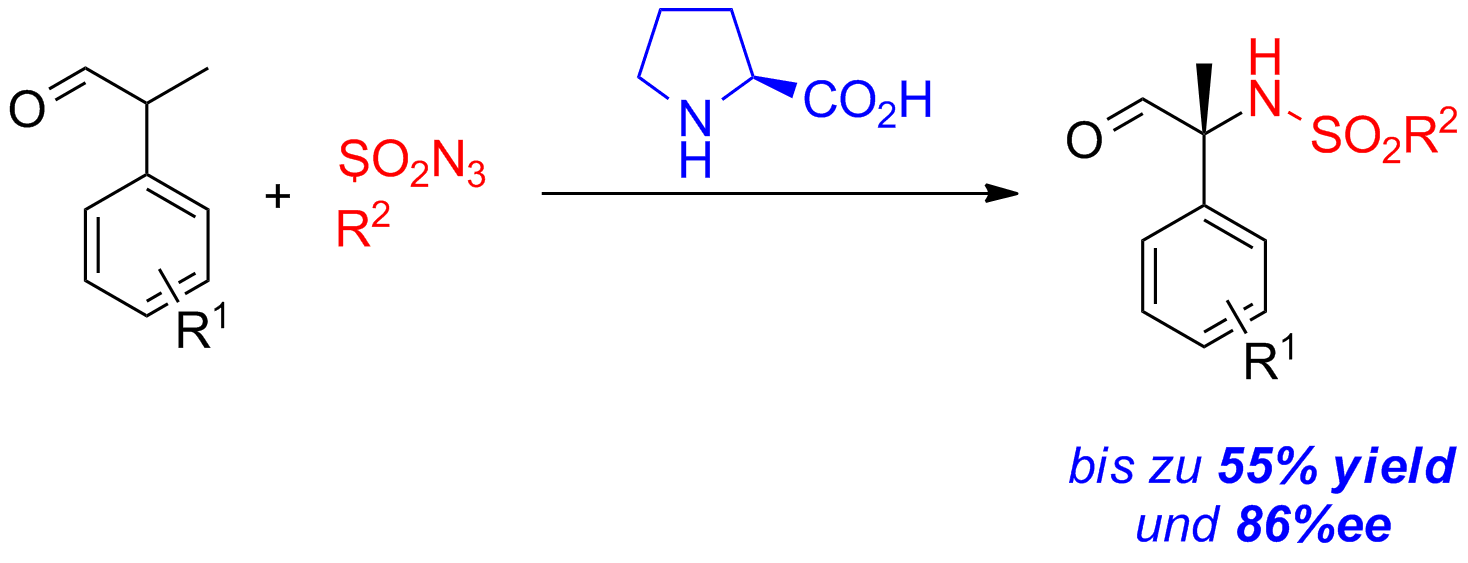
Scheme 2: Asymmetric alpha-sulfamidation of alpha-branched aldehydes.
The obtained products can be transformed with two additional steps into the corresponding unnatural amino acids.
Applying microwave conditions, good to excellent yields under significant reduced reaction times could be obtained. The nitrogen-function can also be introduced through diazo-compounds (DEAD). The α-aminated compounds were obtained in up to 86% ee and moderate to excellent yields (Scheme 3).
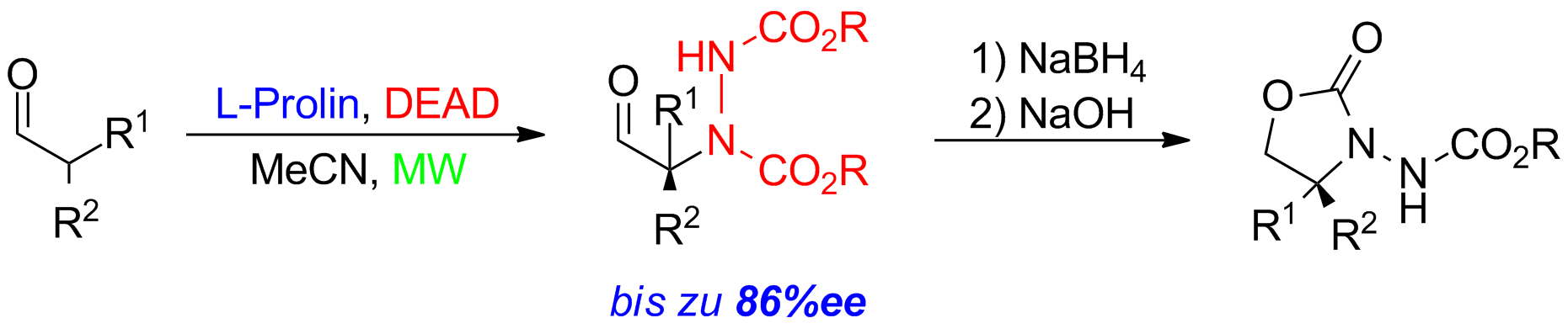
Scheme 3: Proline-catalyzed asymmetric amination of alpha-branched aldehydes.
These products could further be converted both into the corresponding oxazolidinones. But also transformations into α-amino alcohols, carboxylic acids and α-alkylated phenylglycine derivatives are possible.
Asymmetric Diels-Alder-Reaction
The organocatalytic Diels-Alder-reaction is a key-step in the total-synthesis of Δ9-Tetrahydrocannabinol. Various MacMillan-catalysts were tested in this reaction and resulted in high enantioselectivities up to 98% (Scheme 4).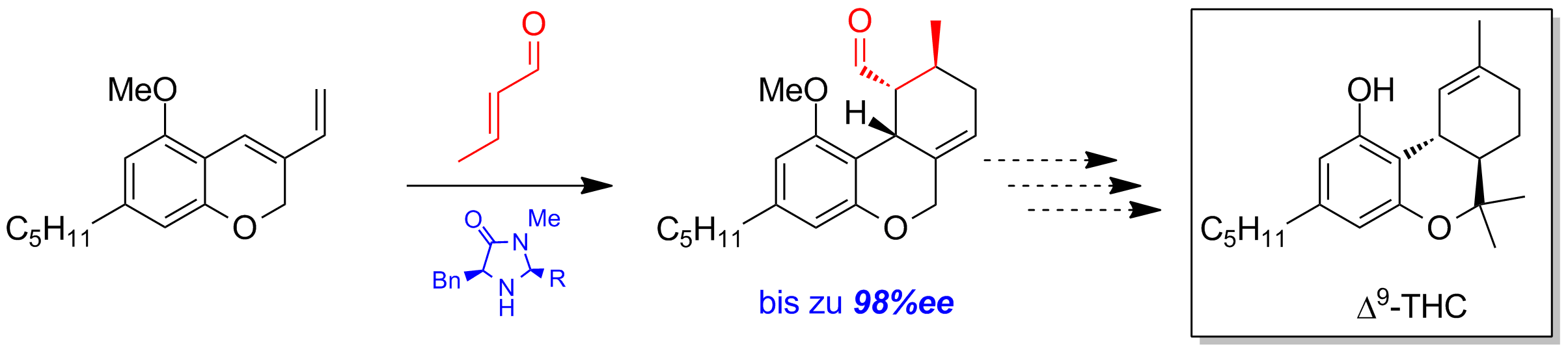
Scheme 4: Asymmetric Diels-Alder with MacMillan-catalysts.
Domino Oxa-Michael-Aldol-Reaction / Formal totalsynthesis of 4-Dehydroxydiversonol
The base-catalyzed domino reaction of salicylaldehyde 1 with α,β-unsaturated aldehydes 2 has been recently investigated in light of their use in total synthesis of natural products. Depending on the reaction conditions in most cases an oxa-Michael-aldol reaction occurs to give chromenes 3. However, under a different set of conditions we and others discovered that rigid tricyclic systems 4 are formed by a vinylogous aldol-oxa-Michael reaction (Scheme 5).
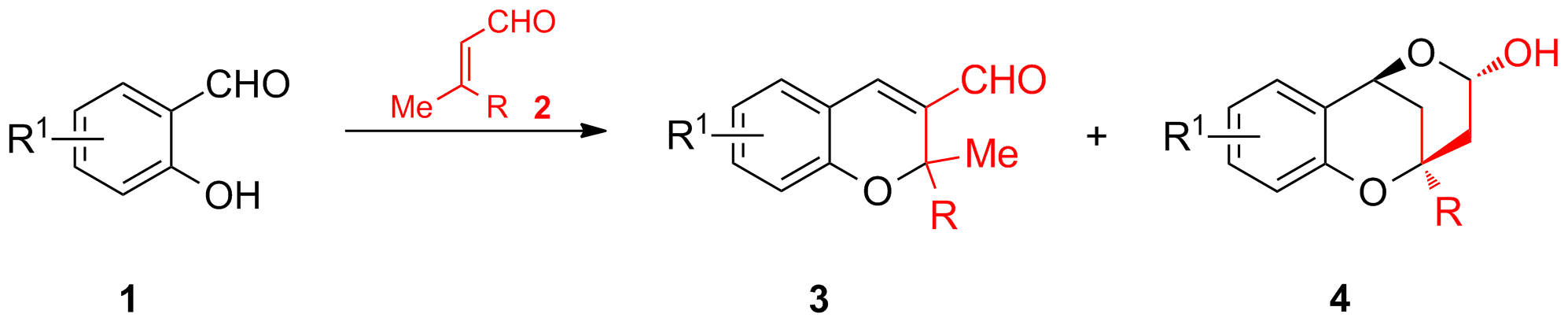
Scheme 5: Possible products of the Domino Oxa-Michael-Aldol-Reaction.
With this method in hand, we converted salicylaldehyde 1a (available from orcinol over 3 steps) to the lactol 4a in high enantiopurities using Jørgensen’s proline-derived organocatalyst 5. Lactol 4a can be transformed with additional steps into xanthone derivative 6, which finally delivers 4-Dehydroxydiversonol (7) according to Tietze et al. (Scheme 6).
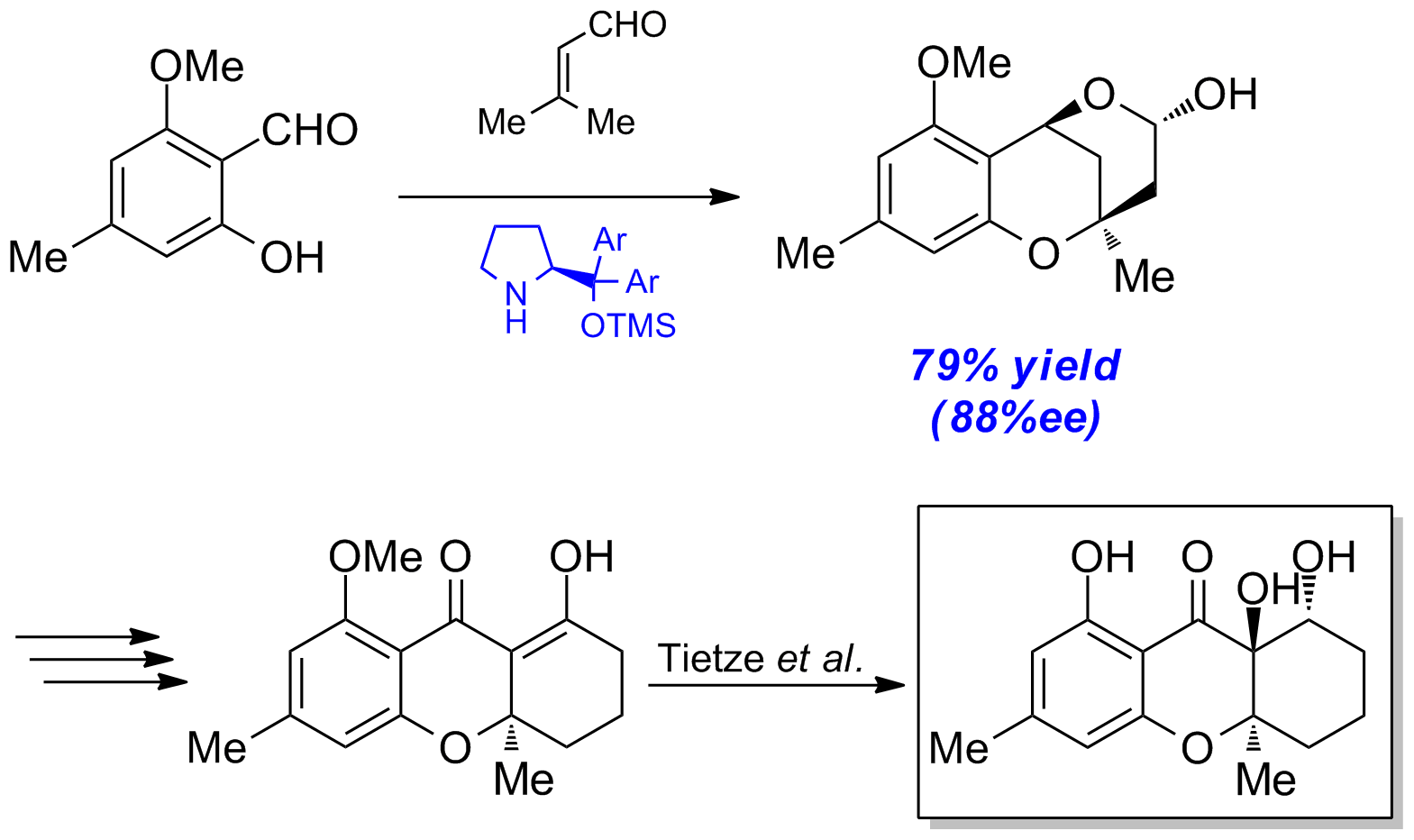
Metal-Catalysis
Asymmetric Synthesis using a Rh-PCP catalyst
Transition metal catalyzed decomposition of diazo compounds offers an easy, scalable, and versatile tool to access cyclopropanes.[11] Even though, rhodium carboxylate complexes are the most attractive catalysts in terms of efficiency and accessibility for the construction of cyclopropane and cyclopropene rings, a major limitation remains their lack of selectivity over b-hydride migration.[12]
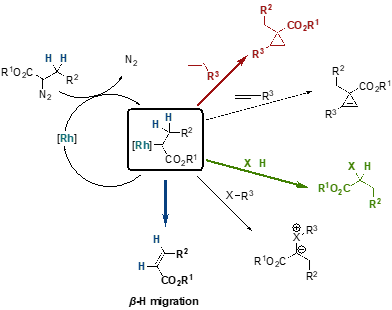
Scheme 7: Possible reactions and mechanism.
[2.2]Paracyclophane carboxylic acid and derivatives thereof are synthesized. The corresponding rhodium paddlewheel complex is applied in the cyclopropanation reaction of terminal, a,a -, and a,b -disubstituted alkenes with a-alkyl-a-diazo esters. The unique bulkiness of [2.2]paracyclophane allows for a chemoselective high yielding cyclopropanation to occur at room temperature, while its inherent planar chirality is currently investigated in enantioselective cyclopropanation reactions.[13]
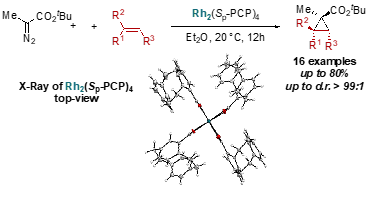
Scheme 8: Tested reactions.
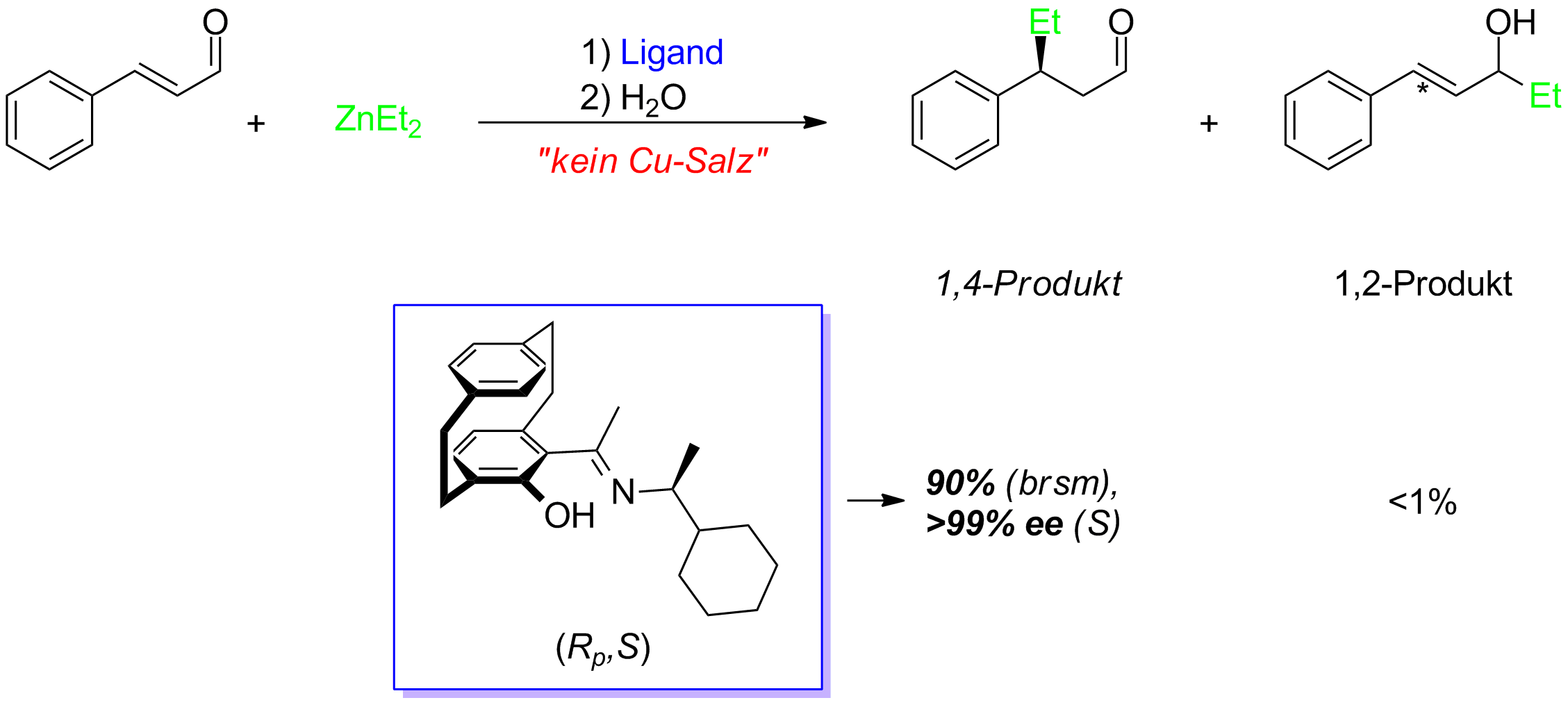
Asymmetric Michael-Addition
Under Cometal-free reaction conditions α,β-unsaturated aldehydes can be used as starting materials in an asymmetric Michael-addition. For this purpose only 2 mol% of the N,O-ligand based on [2.2]paracyclophane are necessary. By this method the desired Michael adducts can be obtained highly selective in excellent enantiomeric excesses (Scheme 7).
Scheme 9: Asymmetric Michael-addition under cometal-free reaction conditions.
References
[1] T. Baumann, H. Vogt, S. Bräse, Eur. J. Org. Chem. 2007, 266–282. The proline-catalyzed asymmetric amination of branched aldehydes.
[2] J. Toräng, S. Vanderheiden, M. Nieger, S. Bräse, Eur. J. Org. Chem. 2007, 943-952. Synthesis of 3-Alkylcoumarins from Salicylaldehydes and α,β-unsaturated Aldehydes utilizing Nucleophilic Carbenes: A New Umpoled Domino Reaction
[3] R. Reingruber, S. Bräse, Chem. Commun. 2008, 105-107. 1,2-Addition of Trialkylaluminium Reagents on N-Diphenylphosphinoyl-ketimines in the Absence of Any Additional Reagents
[4] T. Baumann, M. Bächle, C. Hartmann, S. Bräse, Eur. J. Org. Chem. 2008, 2207-2212. Thermal Effects in the organocatalytic asymmetric α-amination of disubstituted aldehydes with azodicarboxylates: a high-temperature organo-catalysis (Cover picture)
[5] R. Reingruber, S. Vanderheiden, A. Wagner, M. Nieger, T. Muller, M. Es-Sayed, S. Bräse, Eur. J. Org. Chem. 2008, 3314-3327. 1-Aryl-3,3-diisopropyltriazenes: An Easily Accessible and Particularly Stable Class of Triazenes Towards Strong Basic and Lewis Acid Conditions
[6] S. Ay, M. Nieger, S. Bräse, Chem. Eur. J. 2008, 14, 11539-11556. Co-Metal-Free Enantioselective Conjugate Addition Reactions of Zinc Reagents
[7] N. Volz, M. C. Bröhmer, S. Bräse, Synlett 2009, 550-553. An Organocatalytic Sequence towards 4a-Methyl Tetrahydroxanthones: Formal Synthesis of 4-Dehydroxydiversonol
[8] R. Reingruber, S. Vanderheiden, T. Muller, M. Nieger, S. Bräse, Tetrahedron Lett. 2009, in press (invited for spezial issue: 50 years of Tetrahedron publications). Efficient synthesis of 3-acylbenzo[1,2,3]triazenes
[9] R. Reingruber, T. Baumann, S. Dahmen, S. Bräse, Adv. Synth. Catal. 2009, in press. Use of the Chiral Pool – Practical Asymmetric Organocatalytic Strecker Reaction with Quinine
[10] M. C. Bröhmer, N. Volz, S. Bräse, Synlett 2009, in press. Rhodium-catalyzed decarbonylation of functionalized 3-formylchromenes: An (asymmetric) sequence for functionalized chromenes like deoxycordiachromene.
[11] C. Ebner, E. M. Carreira, Chem. Rev. 2017, 117, 11651-11679.
[12] T. L. Sunderland, J. F. Berry, Dalton Trans. 2016, 45, 50-55.
[13] C. Zippel, Z. Hassan, M. Nieger, S. Bräse, Adv. Synth. Catal. 2020.